 What is new is the revelation that the voltage can be tempered to save energy and battery life - without compromising pain relief. The system, which delivers electrical pulses to neutralize pain signals, isn't new. That's the case with new research around Abbott's BurstDR ™ stimulation therapy for chronic pain management. Instead, they're found in incremental improvements. If the medication does not help with pain reduction or if any concerning symptoms occur, the physician should be consulted as soon as possible.Sometimes the biggest innovations don't come as bright and shiny new inventions. All medications must be taken as directed. A combination of pain-relieving medication and antibiotics is usually prescribed. Medications must be taken in a timely manner during the recovery period. When the stimulator is turned on, driving and other activities requiring focus, such as operating machinery should not be performed. Bending, twisting, stretching, and raising the arms above the shoulder level can cause the leads to migrate, so these movements must be restricted as much as possible. Engaging in heavy or strenuous physical activities is discouraged due to the risk of migration or breakage of the leads. Patients are instructed to take a sponge bath and avoid showers, full baths, or water activities. Since the pulse generator device is secured outside the body, care must be taken to not get the device in contact with water. Activity Restrictions During the SCS Trial PeriodĪctivity restrictions help provide better treatment outcomes and protect the device. 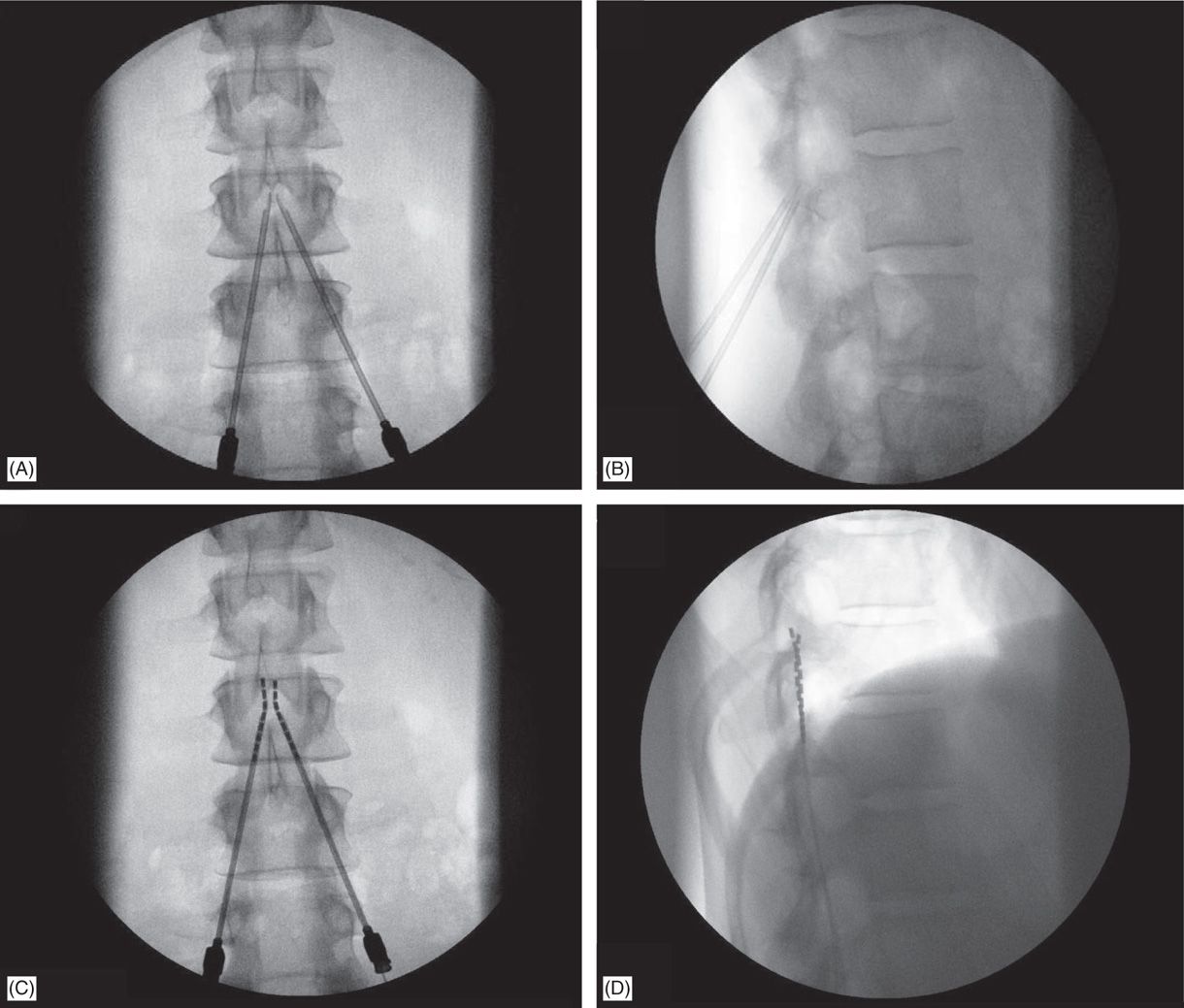
It is advisable to rest for 1-2 days after the insertion of the leads while the body adjusts to the new treatment. Soreness may be treated with ice packs or topical pain medication if approved by the doctor. This setup is temporary, and patients may feel slight discomfort or soreness at the site(s) where the needle was inserted into the skin. Only the leads are inserted into the epidural space. The trial phase of SCS does not include the surgical implantation of the pulse generator. Arranging for a friend or family member to drive is recommended. It is not advisable for patients to drive home after the procedure. The patient is instructed on how to use this device at home for SCS treatment. A manual, handheld device helps program the pulse generator. The pulse generator device is programmed, and the patient is discharged. The patient's vital signs are monitored until the effect of the sedation wears off (about 30 minutes to an hour). The stimulator is about the size of a wallet. 
The outer ends of the leads are connected to a stimulator or pulse generator, which is worn outside the patient’s body. 
The entire procedure is performed under x-ray guidance (fluoroscopy). The procedure involves administering sedation medication to the patient and threading the leads into the spinal epidural space (the area surrounding the spinal cord) through a hollow needle. The entire procedure may take an hour to 90 minutes. The trial is performed as an outpatient procedure in an operating room. What Happens in a Spinal Cord Stimulation Trial In such cases, patients may continue with a permanent implant, if the surgeon approves.Īctivity restrictions apply to both the trial phase and the permanent implant. The goal is to evaluate if the patient experiences at least a 50% reduction in pain and/or 50% improvement in functionality–which is considered a successful trial. Treasure Island (FL): StatPearls Publishing 2022 Jan. The trial is designed to determine the treatment's efficacy and the patient's device tolerance before a permanent implant is done.Ģ Garcia K, Wray JK, Kumar S. 
The SCS trial phase lasts for 3 to 7 days, on average.ġ Dydyk AM, Tadi P. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
